Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Ikeuchi, Hirotomo; Koyama, Shinichi; Osaka, Masahiko; Takano, Masahide; Nakamura, Satoshi; Onozawa, Atsushi; Sasaki, Shinji; Onishi, Takashi; Maeda, Koji; Kirishima, Akira*; et al.
JAEA-Technology 2022-021, 224 Pages, 2022/10
A set of technology, including acid dissolving, has to be established for the analysis of content of elements/nuclides in the fuel debris samples. In this project, a blind test was performed for the purpose of clarifying the current level of analytical accuracy and establishing the alternative methods in case that the insoluble residue remains. Overall composition of the simulated fuel debris (homogenized powder having a specific composition) were quantitatively determined in the four analytical institutions in Japan by using their own dissolving and analytical techniques. The merit and drawback for each technique were then evaluated, based on which a tentative flow of the analyses of fuel debris was constructed.
Walker, C.*
NIMS Bisai Kozo Kaiseki Purattofuomu Riyo Hokokusho (Internet), 2 Pages, 2021/09
High content fly ash silica fume cement (HFSC) has been considering a candidate low alkali cements for the geological disposal of radioactive waste in Japan. JAEA has been currently performing many experiments and modeling studies relevant to validation for the long-term stability of the C-A-S-H gel which is a dominant component of the HFSC. For developing the C-A-S-H model of hydration and degradation, it is necessary to determine the composition of C-A-S-H gel and accompanied minerals when it reacts with water. In the present subject, the synthesized sample of C-A-S-H gels were analyzed by using the NMR spectra of 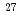 Al and
Al and 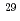 Si to determine the coordination of these element in the C-A-S-H gel.
Si to determine the coordination of these element in the C-A-S-H gel.
Omura, Yoshihito*; Dohi, Terumi
Raiken, 21(2), p.27 - 28, 2021/04
no abstracts in English
Suno, Hiroya; Machida, Masahiko
RIST News, (66), p.3 - 16, 2020/10
no abstracts in English
Kishi, Hirofumi*; Sakamoto, Tomokazu*; Asazawa, Koichiro*; Yamaguchi, Susumu*; Kato, Takeshi*; Zulevi, B.*; Serov, A.*; Artyushkova, K.*; Atanassov, P.*; Matsumura, Daiju; et al.
Nanomaterials (Internet), 8(12), p.965_1 - 965_13, 2018/12
Times Cited Count:11 Percentile:48.71(Chemistry, Multidisciplinary)Hayashida, Kazuki; Kato, Toshihiro*; Kubota, Mitsuru*; Murakami, Hiroaki; Amano, Yuki; Iwatsuki, Teruki
Chikyu Kagaku, 52(1), p.55 - 71, 2018/03
In this study, the simulated experimental drift was constructed in the granite of 500 m depth at Mizunami Underground Research Laboratory, and the hydrochemical process after the drift closure was observed. The groundwater chemistry around the drift changed with the change of the groundwater flow in the fractures when the gallery was constructed. The redox potential increased due to the infiltration of oxygen from the drift into the rock. After closing the drift, the redox potential of the groundwater plunged due to microbial activity, while the groundwater became alkalized conditon due to the influence of cement material such as shotcrete. The amount of cement material consumed for this alkalization was small, and it was considered that its influence would last long in accordance with the amount of cement used.
Seno, Yasuhiro*; Nakayama, Masashi; Sugita, Yutaka; Tanai, Kenji; Fujita, Tomoo
JAEA-Data/Code 2016-011, 164 Pages, 2016/11
The cementitious materials are used as candidate materials for the tunnel support of the deep geological repository of high-level radioactive wastes (HLW).Generally the pH of leachate from concrete mixed Ordinary Portland Cement (OPC) shows a range of 12 to 13. The barrier function of bentonite used as a buffer material and that of host rock might be damaged by the high alkaline leachate from cementitious materials. Therefore, low alkalinity that does not damage each barrier function is necessary for cementitious materials used for the tunnel support system of the HLW geological repository. JAEA has developed a low alkaline cement named as HFSC (Highly Fly-ash contained Silicafume Cement) which the pH of the cement leachate could lower approximately 11. We have confirmed the applicability of HFSC for the tunnel support materials, by using experimentally as the shotcreting materials to the part of gallery wall at 140m, 250m and 350m depth in Horonobe Underground Research Laboratory. And moreover, HFSC has been used as the cast-in-place concrete for the shaft lining concrete at the depth of 374m-380m. This Data/Code summarized the past HFSC mix proportion test results about the fresh concrete properties and hardened concrete properties, in order to offer the information as a reference for selecting the mix proportion of HFSC concrete adopted to the disposal galleries et al. in the future.
Baba, Yuji; Shimoyama, Iwao; Hirao, Norie*
Applied Surface Science, 384, p.511 - 516, 2016/10
Times Cited Count:5 Percentile:25.78(Chemistry, Physical)Chemical states of cesium as well as the other alkali metals (sodium and rubidium) sorbed in micaceous oxides have been investigated by total reflection X-ray photoelectron spectroscopy (TR-XPS). For cesium, it was shown that ultra-trace amount of cesium down to about 100 pg/cm can be detected by TR-XPS. This amount corresponds to about 200 Bq of
can be detected by TR-XPS. This amount corresponds to about 200 Bq of  Cs (t
Cs (t = 30.2y). It was demonstrated that ultra-trace amount of cesium corresponding to radioactive cesium level can be measured by TR-XPS. As to the chemical states, it was found that core-level binding energy in TR-XPS for trace-level cesium shifted to lower-energy side compared with that for thicker layer. A reverse tendency is observed in sodium. Based on charge transfer within a simple point-charge model, it is concluded that chemical bond between alkali metal and micaceous oxide for ultra-thin layer is more polarized that for thick layer.
= 30.2y). It was demonstrated that ultra-trace amount of cesium corresponding to radioactive cesium level can be measured by TR-XPS. As to the chemical states, it was found that core-level binding energy in TR-XPS for trace-level cesium shifted to lower-energy side compared with that for thicker layer. A reverse tendency is observed in sodium. Based on charge transfer within a simple point-charge model, it is concluded that chemical bond between alkali metal and micaceous oxide for ultra-thin layer is more polarized that for thick layer.
Seno, Yasuhiro*; Noguchi, Akira*; Nakayama, Masashi; Sugita, Yutaka; Suto, Shunkichi; Tanai, Kenji; Fujita, Tomoo; Sato, Haruo*
JAEA-Technology 2016-011, 20 Pages, 2016/07
Cementitious materials are expected to be used for the construction of an underground repository for the geological disposal of radioactive wastes. Ordinary Portland Cement(OPC) would conventionally be used in the fields of civil engineering and architecture, however, OPC has the potential to generate a highly alkaline plume (pH 12.5), which will likely degrade the performance of other barriers in the repository such as the bentonite buffer and/or host rock. Low alkaline cementitious materials are therefore being developed that will mitigate the generation of a highly alkaline plume. JAEA has developed a High-volume Fly ash Silica fume Cement (HFSC) as a candidate low alkaline cementitious material. The workability of the HFSC shotcrete was confirmed by conducting In-situ full scale construction tests in the Horonobe underground research laboratory. This report summarizes the results of immersion tests to assess the long-term pH behavior of hardened HFSC cement pastes made with mix designs that are expected to be able to be used in the construction of an underground repository in Japan.
12.5), which will likely degrade the performance of other barriers in the repository such as the bentonite buffer and/or host rock. Low alkaline cementitious materials are therefore being developed that will mitigate the generation of a highly alkaline plume. JAEA has developed a High-volume Fly ash Silica fume Cement (HFSC) as a candidate low alkaline cementitious material. The workability of the HFSC shotcrete was confirmed by conducting In-situ full scale construction tests in the Horonobe underground research laboratory. This report summarizes the results of immersion tests to assess the long-term pH behavior of hardened HFSC cement pastes made with mix designs that are expected to be able to be used in the construction of an underground repository in Japan.
Kurosaki, Yuzuru
Shototsu, 12(4), p.114 - 125, 2015/07
no abstracts in English
Tanaka, Tadao; Sakamoto, Yoshifumi; Yamaguchi, Tetsuji; Takazawa, Mayumi; Akai, Masanobu; Negishi, Kumi; Iida, Yoshihisa; Nakayama, Shinichi
JAERI-Conf 2005-007, p.105 - 110, 2005/08
Highly alkaline environments induced by cementitious materials in radioactive waste repositories are likely to dissolve and to alter montmorillonite, the main constituent of bentonite buffer materials. For the prediction of the long-term variations in permeability of compacted sand-bentonite mixtures, long-term alteration of bentonite should be quantified based on information accumulated by using the compacted or powdered bentonite materials, with batch experiments or column experiments. In this study, we summarize distinctive information obtained from various experimental systems, and propose functional and effective integration of experimental approaches to prediction of bentonite alteration.
Watanabe, Takeshi*; Tsushima, Satoru*; Yamamoto, Ichiro*; Tomioka, Osamu; Meguro, Yoshihiro; Nakashima, Mikio; Wada, Ryutaro*; Nagase, Yoshiyuki*; Fukuzato, Ryuichi*
Proceedings of 2nd International Symposium on Supercritical Fluid Technology for Energy and Environment Applications (Super Green 2003), p.363 - 366, 2004/00
Recovery of salts by supercritical fluid leaching (SFL) method using carbon dioxide was experimentally studied. It was confirmed that LiCl was recovered with a mixed fluid of carbon dioxide and methanol, and KCl and SrCl were recovered with a mixed fluid of carbon dioxide, methanol and crown ether. The influence of crown ether for KCl and SrCl
were recovered with a mixed fluid of carbon dioxide, methanol and crown ether. The influence of crown ether for KCl and SrCl extraction were found to increase in the order of 15-crown-5 (15C5)
extraction were found to increase in the order of 15-crown-5 (15C5)  18-crown-6 (18C6)
18-crown-6 (18C6)  dicychlohexyl-18-crown-6 (DC18C6). It is expected that other salts can be recovered selectively with a mixed fluid of carbon dioxide, methanol and suitable crown ether.
dicychlohexyl-18-crown-6 (DC18C6). It is expected that other salts can be recovered selectively with a mixed fluid of carbon dioxide, methanol and suitable crown ether.
Nagase, Yoshiyuki*; Masuda, Kaoru*; Wada, Ryutaro*; Yamamoto, Ichiro*; Tomioka, Osamu; Meguro, Yoshihiro; Fukuzato, Ryuichi*
Proceedings of 2nd International Symposium on Supercritical Fluid Technology for Energy and Environment Applications (Super Green 2003), p.254 - 257, 2004/00
no abstracts in English
Seko, Noriaki; Katakai, Akio; Tamada, Masao; Sugo, Takanobu*; Yoshii, Fumio
Separation Science and Technology, 39(16), p.3753 - 3767, 2004/00
Times Cited Count:83 Percentile:90.32(Chemistry, Multidisciplinary)Fibrous amidoxime adsorbents were prepared by radiation-induced co-grafting of acrylonitrile (AN) and methacrylic acid (MAA) and subsequent amidoximation. Adsorption of uranium in seawater was evaluated by pumping seawater into the adsorbent column. The best monomer ratio of AN and MAA was 7:3 for continual usage of uranium adsorption. Though hydrochloric acid is an effective eluting agent for the metals adsorbed on the adsorbent, amidoxime groups were simultaneously damaged after five cycles of adsorption-desorption. This deterioration was reduced by an alkaline treatment of the adsorbents after each elution. Furthermore, various organic acids were examined as elution agents. It was found that the 80% of adsorption activity was still maintained after five cycles of adsorption-desorption when tartaric acid was used for eluting agent.
Tachimori, Shoichi; Suzuki, Shinichi; Sasaki, Yuji; Apichaibukol, A.
Solvent Extraction and Ion Exchange, 21(5), p.707 - 715, 2003/09
Times Cited Count:48 Percentile:75.59(Chemistry, Multidisciplinary)no abstracts in English
 C=C:) from charge inversion mass spectrometry
C=C:) from charge inversion mass spectrometryHayakawa, Shigeo*; Tomozawa, Koji*; takeuchi, takae*; Arakawa, Kazuo; Morishita, Norio
Physical Chemistry Chemical Physics, 5(11), p.2386 - 2390, 2003/05
Times Cited Count:15 Percentile:44.34(Chemistry, Physical)The process of formation of the negative ions in charge inversion mass spectrometry is via near-resonant neutralization followed by spontaneous dissociation, and then endothermic negative ion formation. The intensity of the C
 peak relative to the C
peak relative to the C H
H peak in these spectra increased with decreasing ionization potential of the targets for both of the isomeric C
peak in these spectra increased with decreasing ionization potential of the targets for both of the isomeric C H
H
 cations. The formation of the C
cations. The formation of the C
 anion is proposed to result from the dissociation of excited C
anion is proposed to result from the dissociation of excited C H
H neutrals into C
neutrals into C and H
and H . The dependence on target species of the intensities of C
. The dependence on target species of the intensities of C
 peak relative to the C
peak relative to the C H
H peak for HCCH and H
peak for HCCH and H C=C: cannot be rationalized by the internal energy of the excited C
C=C: cannot be rationalized by the internal energy of the excited C H
H neutrals. The differences indicate that the isomeric C
neutrals. The differences indicate that the isomeric C H
H neutrals dissociate into C
neutrals dissociate into C H and H prior to 1,2-hydrogen atom migration.
H and H prior to 1,2-hydrogen atom migration.
Taniguchi, Naoki; Kawakami, Susumu; *
JNC TN8400 2001-025, 27 Pages, 2002/03
It is essential to understand the corrosion type of carbon steel under the repository conditions for the lifetime assessment of carbon steel overpack used for geological isolation of high-level radioactive waste. According to the previous study, carbon steel is hard to passivate in buffer material assuming a chemical condition range of groundwater in Japan. However, concrete support will be constructed around the overpack in the case of repository in the soft rock system and groundwater having a higher pH may infiltrate to buffer material. There is a possibility that the corrosion type of carbon steel will be influenced by the rise of the pH in groundwater. In this study, anodic polarization experiments were performed to understand the passivation condition of carbon steel in buffer material saturated with water contacted with concrete. An ordinary concrete and a low-alkalinity concrete were used in the experiment. The results of the experiments showed that the carbon steel can passivate under the condition that water having pH  13 infiltrate to the buffer material assuming present property of buffer material. If the low-alkalinity concrete is selected as the support material, passivation can not occur on carbon steel overpack. The effect of the factors of buffer material such as dry density and mixing ratio of sand on the passivation of carbon steel was also studied. The results of the study showed that the present property of buffer material is enough to prevent passivation of carbon steel.
13 infiltrate to the buffer material assuming present property of buffer material. If the low-alkalinity concrete is selected as the support material, passivation can not occur on carbon steel overpack. The effect of the factors of buffer material such as dry density and mixing ratio of sand on the passivation of carbon steel was also studied. The results of the study showed that the present property of buffer material is enough to prevent passivation of carbon steel.
*; Mihara, Morihiro;
JNC TN8430 2001-007, 56 Pages, 2002/01
In the geological disposal concept of radioactive wastes, a kind of clay with sorption ability and low permeability, called bentonite, is envisaged as an engineered barrier system in the geological repository. Also, the cemetitious material is envisaged as the backfill material in the vaults and the structure material of the vaults. The groundwater in contact with the cementitious material will promote hyperalkaline conditions in the repository environment and these conditions will affect the performance of the bentonite. Therefore, it is necessary to investigate the interaction between the cementitious material and the bentonite for the evaluation of long term stability of the disposal system. In this study, for the identification and the investigation of the secondary minerals, the batch immersion experiments of the powder bentonite were carried out using synthetic cement leachates (pH=7, 12.5, 14) at 200 C. As the results, it was confirmed that Na as exchangeable cations in the bentonite can exchange relatively easily with Ca in the solution from the experiment results. And the ratio of cation exchange was estimated to be about 25% based on the amount of exchangeable cations Ca
C. As the results, it was confirmed that Na as exchangeable cations in the bentonite can exchange relatively easily with Ca in the solution from the experiment results. And the ratio of cation exchange was estimated to be about 25% based on the amount of exchangeable cations Ca between layers. Furthermore, it was concretely shown that the generation of analcime might be affected by the Na concentration from results of the solution analyses and a stability analysis of analcime using the chemical equilibrium model, in addition to the pH in the solution.
between layers. Furthermore, it was concretely shown that the generation of analcime might be affected by the Na concentration from results of the solution analyses and a stability analysis of analcime using the chemical equilibrium model, in addition to the pH in the solution.
Apel, P. Y.*; Korchev, Y. E.*; Siwy, Z.*; Spohr, R.*; Yoshida, Masaru
Nuclear Instruments and Methods in Physics Research B, 184(3), p.337 - 346, 2001/11
Times Cited Count:477 Percentile:99.96(Instruments & Instrumentation)no abstracts in English
Fukumoto, Masahiro; Nishikawa, Yoshiaki*
JNC TN8400 2001-017, 355 Pages, 2001/03
The alkali hydrolysis experiments which seem to be important from the view point of the alteration mechanism using the following seven organic materials was performed as a part of the evaluation of the influence on the disposal of the organic materials contained in the TRU wastes. As a result of the alkali hydrolysis experiments (90 C and 91d), each organic materials became those of lower molecular weight. The degradation products were able to be detected in the solution. The organic materials seem to be degraded to the organic matters which were confirmed in this study in a long term of disposal. The degradation products were shown below. Therefore, the evaluation of the influence on the migration of radionuclides by degradation products becomes important in the future. (1)Cement additives of Naphthalenesulfonic acid and Ligninsulfonic acid (
C and 91d), each organic materials became those of lower molecular weight. The degradation products were able to be detected in the solution. The organic materials seem to be degraded to the organic matters which were confirmed in this study in a long term of disposal. The degradation products were shown below. Therefore, the evaluation of the influence on the migration of radionuclides by degradation products becomes important in the future. (1)Cement additives of Naphthalenesulfonic acid and Ligninsulfonic acid ( Naphthalenedisulfonic acid etc.) (2)Cement additives of polycarboxylic acid (
Naphthalenedisulfonic acid etc.) (2)Cement additives of polycarboxylic acid ( Oligomer of distal methoxypoly ethylene glycol.) (3)Ethylenediamine-N,N,N',N'-tetraacetic acid disodium salt (
Oligomer of distal methoxypoly ethylene glycol.) (3)Ethylenediamine-N,N,N',N'-tetraacetic acid disodium salt ( Acetic acid desorped and cyclized organic matters from EDTA.) (4)Tributyl phosphate (
Acetic acid desorped and cyclized organic matters from EDTA.) (4)Tributyl phosphate ( Dibutyl phthalate, n-butanol) (5)Poly vinyl acetate (
Dibutyl phthalate, n-butanol) (5)Poly vinyl acetate ( Acetic acid) (6)Nylon66 (
Acetic acid) (6)Nylon66 ( Adipic acid, Hexamethylenediamine) (7)Cured epoxy resin (
Adipic acid, Hexamethylenediamine) (7)Cured epoxy resin ( Glycerol poly glycidyl ether, Carboxylic acid)
Glycerol poly glycidyl ether, Carboxylic acid)